 Note: ρ is density, n is refractive index at 589 nm, and η is viscosity, all at 20 ☌ T eq is the equilibrium temperature between two phases: ice/liquid solution for T eq < 0–0.1 ☌ and NaCl/liquid solution for T eq above 0.1 ☌. 
For any chemical reaction, the standard entropy change is the sum of the standard molar entropies of the products minus the sum of the standard molar entropies of the reactants. Entropies of gases are much larger than those of condensed phases. The standard molar entropy of a substance is the absolute entropy of 1 mole of the substance in the standard state. The entropy also increases as the pressure or concentration becomes smaller. Thermodynamic properties Phase behaviorĭensity data of aqueous solutions Water–NaCl phase diagram Properties of water–NaCl mixtures NaCl, wt% The entropy of a substance increases with its molecular weight and complexity and with temperature. Combustion of a fuel, once ignited, occurs without any other external influence therefore, it is a spontaneous process, and it occurs at a very high rate. Introduction to entropy Gibbs free energy and thermodynamic favorability. Iron oxide (or rust) is a spontaneous reaction that occurs at a very slow rate in usual atmospheric conditions. 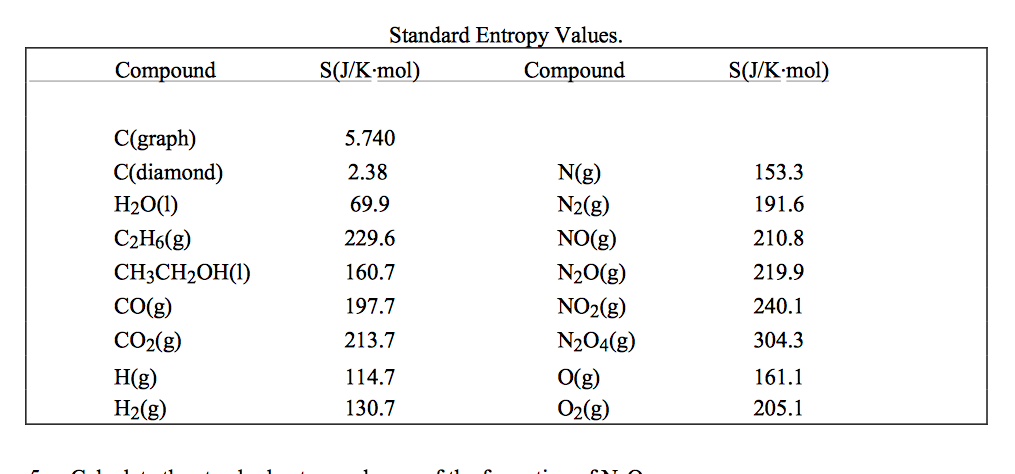
These are often (but not necessarily) chosen to be the standard temperature and pressure. Structure and properties Structure and properties Moles and molar mass Mass spectroscopy of elements Elemental composition of. In chemistry, the standard molar entropy is the entropy content of one mole of pure substance at a standard state of pressure and any temperature of interest. 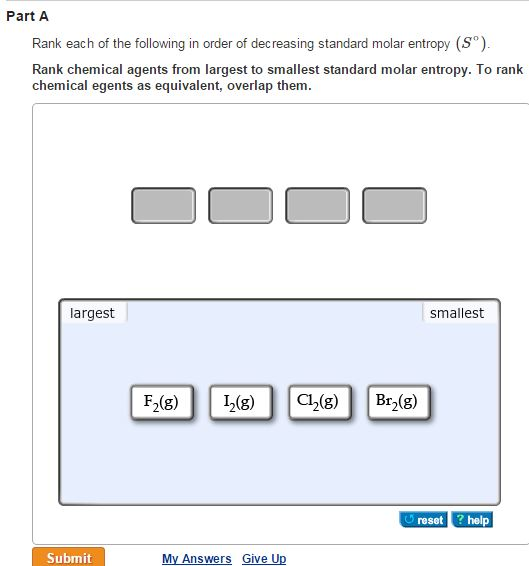
It is highly recommended that you seek the material safety data sheet ( MSDS) for this chemical from a reliable source such as eChemPortal, and follow its direction. The standard state of a substance is an arbitrarily. In ice, molecules of H2O are less random than in liquid water. Standard molar entropy is the amount of entropy one mole of substance has under its standard state. The handling of this chemical may incur notable safety precautions. The standard molar entropy of H2O (l) is 70 JK1 mol1. This page provides supplementary chemical data on sodium chloride. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
